Warning letters increase as DSI rejigs operations
In a press statement issued in August the US Food and Drug Administration (FDA) detailed plans to clampdown on non-compliant investigators and enhance its debarment and disqualification measures.
A spokesperson for the FDA’s Center for Drug Evaluation and Research's (CDER's) Division of Scientific Investigations (DSI) told Outsourcing-Pharma how its operations had changed.
To increase the speed and efficiency of actions against misconduct DSI “staff members have been realigned so that the most serious cases of non-compliance are handled by reviewers dedicated to handling such cases”.
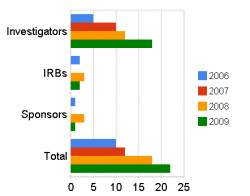
Following this operational shift DSI “has improved its efficiency in issuing warning letters”, according to the spokesperson, and this is supported by the data displayed below which plots the number issued in the past four years.
The increase in 2009 is part of a year-on-year rise in the number of warning letters issued to clinical investigators since 2006. Over this same period letters to institutional review boards (IRB) and clinical trial sponsors have fluctuated and remained low.
Following the US Government Accountability Office’s (GAO) investigation into Coast IRB there have been calls to reform the system in the US. The FDA intends to make IRBs register with the agency and is developing voluntary guidelines.
Data provided to Outsourcing-Pharma by DSI. Figures for 2009 were taken on August 21.














