Oasmia reviewing proposals from firms interested in API solubility enchancement tech

The Swedish firm said it has begun assessing proposals from companies interested in the XR17 technology – which is used to solubilize insoluble active pharmaceutical ingredients (APIs) – but did not respond to a request for more information.
Oasmia also said it planned to ramp up efforts to find marketing and distribution partners for own brand products developed using the technology adding that it has already held talks with “multiple potential partners.”
Phase II data
The news comes weeks after Oasmia released Phase III data showing paclitaxel formulated with XR17 – known variously as Paclical or Apealea – and carboplatin was as effective as Taxol (paclitaxel) and carboplatin in treating patients suffering epithelial ovarian cancer.
Overall survival in the Taxol group was 24.8 months, versus 25.7 months in the group treated with Oasmia’s formulation. At the time Oasmia said the data would form the basis for a US filing by the end of the year.
Apealea was submitted for European Medicines Agency (EMA) review in February based on earlier study data showing it met progression-free survival endpoints.
Differentiation
While the Phase III data are positive, Oasmia has previously stated that XR17 rather than non-inferiority is what really differentiates its product from Taxol.
Taxol is formulated with cremophor EL, a version of polyethoxylated castor oil that is used to stabilize emulsions of nonpolar materials. The excipient, which is sold as Koliphor EL by BASF, has been associated with allergic reactions which means cancer patients treated with Taxol also need treatment with steroids and antihistamines.
According to Oasmia, in the the Phase III study "patients treated with Taxol received systemic pre-treatment with corticosteroids, antihistamines and H2 receptor antagonists" while "patients treated with Paclical did not receive such treatment to the same extent."
Technology and funding
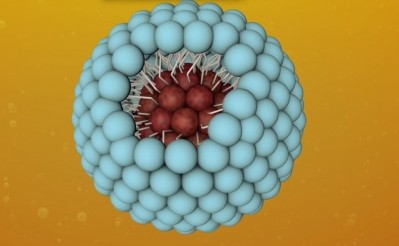
The XR17 excipient is derived from Vitamin A and consists of a polar head and long hydrophobic tail which – when in liquid – forms spherical micelles of approximately 20 to 60nm in diameter.
These micelles can be used to encapsulate insoluble active pharmaceutical ingredients (APIs), rendering them water soluble.
In April, Oasmia raised SEK45.5m ($5.4m) it said it will use to fund operations and the development of additional human and animal drugs that employ the XR17 technology.
At present XR17 is used for five of Oasmia’s cancer candidates including both Apealea and Docecal (docetaxel), which is a potential rival to Sanofi’s blockbuster Taxotere.