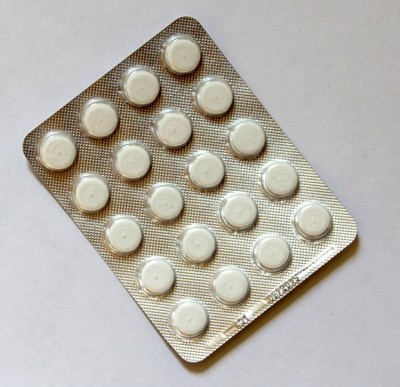
Almac sets blister pack investment in context of dynamic US market
The firm made the prediction weeks after announcing a multimillion dollar investment programme that saw it install a Pharmaworks TF3 at its clinical supply plant in Souderton and a commercial scale Uhlmann UPS 4 at its facility in neighbouring Audubon.
Martin Lamb VP of business development at Almac Clinical Services told Outsourcing-pharma.com that: “We see a movement in the US market away from bottle formats and into blister formats to address issues of patient compliance, brand awareness and combination packs.
He explained that – at present - blister packaging for clinical trials accounts for around 10 per cent of Almac’s contract packaging work and predicted that this is likely to grow as a result of the new investment, particularly the $1m (EUR781,000) it spent on the TF3.
“Our investment in the TF3 widens the scope of clinical blister packaging projects we are able to support in the US by helping our clients to achieve shorter lead times and reduced costs,” Lamb said, adding that Almac’s move to a new HQ gave it the space to expand its US offering.
Commercial scale
The Uhlmann UP4 is – in contrast – designed for commercial scale production according to Almac Pharma Services VP of commercial operations David Downey who told Outsourcing-pharma.com this has cost implications the firm is keeping in mind.
“Operating a line the scale of the UPS 4 does have a significant running cost. However we have specified the line to be as cost effective and flexible as possible, with on-line print capabilities and quick change over.”
Downey also said that the ‘significant client interest’ Almac received prior to the installation of the UP4 “leads us to believe that the investment return will be made quickly.”
Staff training
The investment in the two new technologies was companied by an investment in staff training, with the suppliers providing the contract services firm with full operational support.
“The TF3 machine is user-friendly and with the 2-3 weeks of training and use the machine technician staff are fully knowledgeable on the line” Downey explained.
“The UPS 4 has a significant amount of on-line unit integration, so the training timeline is somewhat extended to approximately 5-6 weeks. However as this line is being sited in our new US commercial packaging facility we have the ability to manage this extended training period within the scope of the overall project timeline.”