Oasmia's vitamin A excipient tech attract global pharma collaborator
Uppsala, Sweden-based Oasmia did not name the pharmaceutical company involved or give financial details of the agreement but did say that the initial focus will be testing if its tech – known as XR-17 – to improve the solubility characteristics of a solid dose formulation.
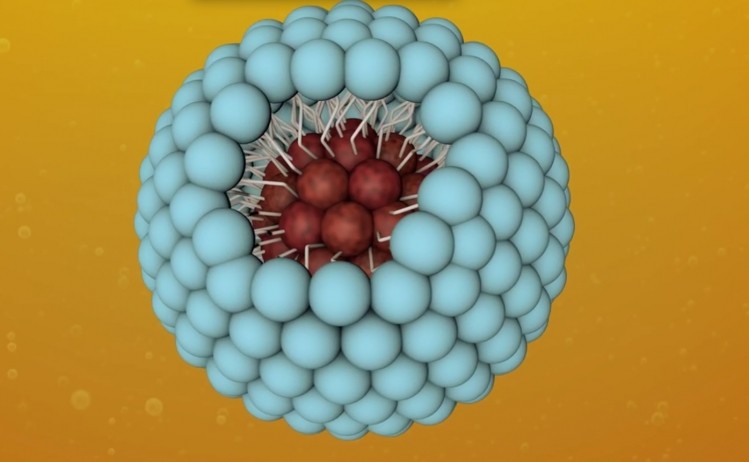
XR-17 is an excipient derived from vitamin A that is used to enhance the solubility of active pharmaceutical ingredients (API). The molecule consists of a polar head and long hydrophobic tail which, when in liquid, form spherical micelles of approximately 20 to 60nm in diameter.
The micelles can be used to encapsulate insoluble APIs, rendering them water soluble.
CEO Julian Aleksov said: "The agreement we have signed today is indeed a validation of the XR-17 technology, which has always been, and continues to be, the heart and soul of Oasmia's research and development.
“With this agreement, we have the opportunity not only to develop additional products within our portfolio but also to add a third leg to our business model apart from our existing human and veterinarian oncology products.”
The XR-17 technology is key to Oasmia’s own portfolio of products –the ovarian cancer drug Paclical and the veterinary paclitaxel formulation Paccal Vet-CA- as well as four other drug candidates currently in development.
CMO contract
News of the deal follows just a day after Oasmia extended its manufacturing contract with Baxter’s BioPharma Solutions division.
The deal, financial terms of which were not made public, is a master manufacturing agreement under which Baxter will produce drug candidates in Oasmia’s development pipeline such as the breast cancer treatment Doxophos.
Oasmia has already contracted Baxter to make commercial supplies of Paclical and Paccal Vet-CA1at its cytotoxics manufacturing facility in Halle, Germany.