Early stage clinical trials get boost from modern EDC platform

“We are a modern EDC (electronic data capture) platform,” said James Crooks, VP of Engineering at the company.
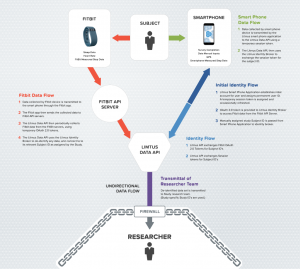
While most EDCs simply route data, Crooks told us Litmus augments data using machine learning and other algorithms, which correlate the data with other environment-based data, such as pollution data, weather-related data, GPS, etc.
The Litmus dashboard displays study’s progress and surfaces population trends, allowing researchers to view individual participants’ data. Additionally, the platform is device agnostic, so it can integrate with various wearables, smart devices, and sensors, regardless of brand or model.
“Another important differentiator is when we sell into the clinical trials process – Phases I and II, instead of Phase III,” explained Crooks.
“Our goal is to provide insight into efficacy and quality of life during toxicity studies so investigators can make informed go/no-go decisions heading into Phase III,” he said, commenting that Phase III is where they see cost “skyrocket and timelines stall.”
If this continues, Crooks said sustainability will depend on “shrewd cost-benefit analysis before initiating Phase III trials.”
However, by informing that decision-making process, Litmus believes it can contribute to a “lean” pharma in a way that Crooks explained would be beneficial to the economics of the industry as a whole.
“Together, the components of the Litmus Health platform allow researchers to collect agile, continuous, multi-stream data from their subjects,” he said. “In turn, these data can be used to make better, more rapid business and clinical decisions.”
All the world’s a clinical trial
Consumers are already generating enormous amounts of data – and in the near future, technology advances will make it simpler to share this data for research purposes, Sam Volchenboum, Chief Science Officer at Litmus told us.
“Everyone will be able to help advance medicine, whether it's by anonymously contributing 23andMe results to a database where researchers look for correlations, opting into answering questions about your prescription’s side effects via a phone app, or donating data from your Fitbit to improve statistical normalization,” he added.
“The more seamless and secure the process, the more we expect to see people sharing their data with the research community, even when they aren’t officially enrolled in a clinical study.”
While the platform currently focuses on the early stage clinical trials, Daphne Kis, Litmus CEO, told us the company plans on focusing on other trial phases. “Any phase of a clinical trial would benefit from better data,” she said.
Echoing Crooks, Kis explained the company started with Phase II and earlier “because of the pain point that the Phase II/III transition represents.”
Currently, Litmus is in talks with several customers, and Kis said to expect announcements in the next four to six months on that front. The company is also expanding into other disease areas.
“We truly do believe that pharma has a noble purpose at its core, which is to develop treatments to cure or manage diseases, to help people live longer and better, and to better understand the human body,” said Kis.
“However, the prevalence of inefficiency, waste, and poor data in clinical trials makes this hard to achieve,” she said.
The platform is currently being piloted at the University of Chicago in a clinical trial run by gastroenterologist Dr. David Rubin on the effects activity, sleep, and diet on Inflammatory Bowel Disease (IBD) patients.